 
Electrons move around outside the nucleus.The nucleus carries a positive electrical charge. 
The nucleus of an atom contains protons and neutrons.The mass of a proton is 1840 times greater than the mass of an electron. The mass of a proton is essentially the same as that of a neutron.Protons and neutrons are about the same size as each other and are much larger than electrons.In other words, neutrons do not have a charge and are not electrically attracted to either electrons or protons. Electrons and protons are electrically attracted to each other. The charge of a proton and an electron are equal in magnitude, yet opposite in sign. Each proton has a positive electrical charge.Each electron has a negative electrical charge.They do consist of parts, which include protons, neutrons, and electrons, but an atom is a basic chemical building block of matter. Atoms cannot be divided using chemicals. 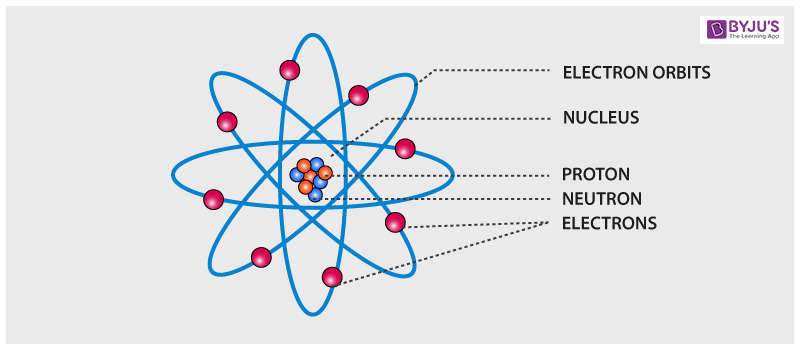
Protons and neutrons are found together in what is called the nucleus of the atom. Electrons have a negative electrical charge. Protons have a positive electrical charge. An atom consists of three main parts: protons, neutrons, and electrons. The fundamental building block of matter is the atom. An atom with too many or too few electrons is unstable and may bond with another atom to either share or essentially donate electrons.Ĭhemistry is the study of matter and the interactions between different types of matter and energy.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
